The ISO/IEC 17025 Testing and Calibration Laboratories Implementation Toolkits provide a comprehensive, ready-to-use suite of premium templates and practical resources to help laboratories establish, implement, maintain, and continually improve management system and technical competence requirements with consistency, precision, and operational confidence.
Aligned with ISO/IEC 17025 requirements, this
toolkit translates laboratory quality, impartiality, and technical competence
expectations into actionable policies, procedures, forms, records, and
monitoring tools enabling organizations to strengthen laboratory governance,
improve the reliability of testing and calibration results, enhance audit and
accreditation readiness, and support sustained performance across laboratory
operations.
|
Price: $296.00

Secure payment via PayPal. Accepted methods include PayPal and major credit cards.
✔ Instant Download | ✔ Secure Payment | ✔ No Subscription
|
Below is a list of documents you will find in the package. Click on index file button to see which contents are included.
|
|
| Date File Updated | 25/03/2025 |
| File Format | pdf, xls, doc |
| No. of files | 191 Files, 16 Folders |
| File download size | 5.50 MB (.rar) |
| Language |
|
| Purchase code | ISO17025-Toolkits |
|
This document has been certified by a professional 100% customizable. You can edit our templates as needed. Instant download after completing your order. Our download process takes less than 2 minutes. We recommend downloading this file onto your computer. Your payment information is processed securely. After payment, if you require an invoice, please email us. Support contact: supports@iso-toolkits.org |
|
1. Who are these ISO toolkits designed for?
The ISO toolkits available on ISO-Toolkits.org are specifically designed for quality, compliance, risk, operations, and management system professionals who are responsible for implementing, maintaining, auditing, or improving ISO standards within an organization, including but not limited to:
Quality Managers and QMS Coordinators
ISO Implementation Managers and Management Representatives
Compliance Officers and Risk Management Professionals
Internal Auditors and Lead Auditors
Operations, HSE, HR, IT, Security, and Production Managers
Consultants supporting ISO certification, surveillance audits, and management system improvement projects
Corporate trainers, lecturers, and professional training providers delivering ISO-related programs
These ISO toolkits are suitable for:
Manufacturing, construction, logistics, healthcare, education, IT, services, public sector, and other industries implementing ISO-based management systems
Small and medium-sized enterprises (SMEs) preparing for certification with limited internal documentation resources
Large enterprises and multi-site organizations seeking standardized documents, audit tools, and implementation frameworks
Independent consultants and training institutions in need of structured, editable, and ready-to-use ISO implementation materials
2. What does each ISO toolkit include?
Each ISO toolkit on ISO-Toolkits.org is built as a comprehensive, practical, and structured implementation package tailored to real-world organizational use. Depending on the standard or topic (ISO 9001, ISO 14001, ISO 45001, ISO 27001, ISO 22000, ISO 13485, ISO 22301, integrated management systems, etc.), a typical toolkit includes:
🔹 1. Management System Templates (Word files)
Editable policies, procedures, process maps, forms, registers, plans, checklists, work instructions, and audit reports
Organized by ISO clause, process area, department, or implementation phase for easy navigation
Fully customizable to match your organization's scope, industry, risk profile, and internal processes
🔹 2. Audit, Risk & Performance Spreadsheets (Excel files)
Risk registers, compliance matrices, audit schedules, nonconformity logs, corrective action trackers, KPI dashboards, and scoring tools
Equipped with formulas, tables, filters, charts, and tracking fields to support implementation and monitoring
🔹 3. Professional Presentation Slides (PowerPoint files)
Ready-to-use slides for ISO awareness training, management briefings, implementation kickoffs, and internal communication
Professionally designed and brand-customizable for internal or client-facing use
🔹 4. User Guides & Implementation Notes
Practical guidance explaining the purpose, usage, and implementation sequence of each document group
Recommendations on adapting the materials based on industry, business size, certification scope, and organizational maturity
3. How many templates/documents are included in each ISO toolkit?
The number of documents varies by toolkit, but most ISO toolkits are developed as comprehensive, in-depth packages covering core implementation, operation, monitoring, auditing, and improvement requirements.
✅ Single ISO Standard Toolkits:
80-200 Word templates, depending on the standard, industry scope, and level of detail
20-60 Excel tools such as risk registers, audit trackers, compliance matrices, KPI dashboards, and action logs
10-40 PowerPoint presentations for training, awareness, leadership communication, and implementation planning
✅ Integrated Management System Toolkits:
150-300+ documents covering multiple standards such as ISO 9001, ISO 14001, ISO 45001, ISO 27001, or other integrated requirements
Templates designed to reduce duplication and support one unified management system across departments
✅ Industry-Specific ISO Toolkits:
Specialized documents adapted for sectors such as manufacturing, healthcare, food, construction, education, IT services, logistics, and public administration
4. Can I preview the content before purchasing?
Yes. We understand that reviewing ISO documentation before purchase is important for informed decision-making, especially when preparing for certification, internal audits, or management system implementation. Therefore, on each toolkit's product page, we provide:
Sample screenshots of templates, audit tools, dashboards, or training slides
Full list of included files with module names, document categories, and file IDs
For selected ISO toolkits, free downloadable samples or sample previews upon request
If you require a preview of specific documents, clauses, forms, or implementation modules before purchasing, feel free to contact us via email or the website form. Our support team will respond promptly with tailored assistance.
5. Are these ISO toolkits suitable for small and medium-sized businesses (SMEs)?
Absolutely. The ISO toolkits are built with flexibility, scalability, and practical usability, making them suitable not only for large enterprises but also for SMEs that are:
Building a management system for the first time
Preparing for ISO certification, recertification, or surveillance audits
Standardizing processes, forms, records, and internal controls across departments
Seeking practical, ready-to-use materials without large consulting budgets
✅ Key benefits for SMEs:
Easy-to-use templates that can be selectively applied based on your certification scope
No complex systems required - just Word, Excel, and PowerPoint
Significant cost and time savings compared to developing ISO documentation from scratch
Step-by-step guidance to empower internal teams, process owners, and management representatives
6. What file formats are used in the ISO toolkits? (Word, Excel, PowerPoint?)
All documents are provided in fully editable, standard office formats, compatible with widely used software such as Microsoft Office and Google Workspace.
✅ Supported file types:
Microsoft Word (.docx):
For policies, procedures, process descriptions, SOPs, work instructions, forms, audit reports, management review records, and ISO documentation.
→ Easy to edit, insert content, and tailor to your organizational structure.Microsoft Excel (.xlsx):
For risk registers, compliance matrices, internal audit plans, corrective action trackers, KPI dashboards, process performance sheets, and scoring tools.
→ Built-in formulas, charts, filters, and conditional formatting included.Microsoft PowerPoint (.pptx):
For ISO awareness training, implementation kickoffs, management briefings, process owner workshops, and internal communication slides.
→ Professionally designed and ready for brand customization.
7. Are the templates editable?
Yes. All templates are 100% editable.
They are delivered in original, editable formats (Word, Excel, PowerPoint), giving users full flexibility to:
Customize the content to meet specific ISO requirements, business needs, process flows, responsibilities, and internal terminology
Insert company logo, brand elements, document codes, approval fields, and internal policies
Adjust layouts, colors, languages, and document control information to fit your organization's format and management system style
Apply across departments, sites, processes, projects, or branches with full flexibility
8. Are ISO toolkit contents regularly updated? What is the update policy?
Yes. We regularly update ISO toolkits to keep pace with changes in international standards, audit practices, regulatory expectations, industry requirements, and practical implementation methods.
🔄 Update policy:
All customers are notified by email when new updates are released
Minor updates (e.g., error corrections, improved instructions, minor enhancements, format improvements, additional usage notes) are free within 6-12 months of purchase (depending on the product)
Major updates (e.g., alignment with revised ISO versions, new clause interpretation, major content restructuring, additional modules, or expanded industry-specific templates) will be offered to previous customers at exclusive upgrade discounts
📌
Recommendation:
Keep your order confirmation email
and reference ID to easily access
update privileges in the future.
🎯 Our commitment is to ensure customers always have access to modern, practical, and audit-ready ISO implementation tools to support successful certification and continual improvement.
9. Can I use the templates immediately, or do I need to adjust them first?
You can start using them right away. All templates are designed based on ISO management system requirements, implementation good practices, and real-world documentation structures for immediate deployment.
✅ Key features:
Built with practical ISO implementation content and standard-aligned structures
Come with usage instructions and practical context for management system deployment
Organized by ISO clauses, management system processes, or implementation phases for step-by-step or full-system deployment
However, to maximize relevance, we recommend:
✏️ Adjusting certain fields (e.g., company name, process owners, scope, risks, KPIs, document codes, approval roles)
✏️ Localizing terminology, formatting, language, legal references, and operational details based on your industry and certification scope
🎯 With their professional design and ready-to-use content, the toolkits help you reduce 60-80% of documentation time while improving consistency, audit readiness, and implementation quality.
10. Do ISO toolkits come with user guides or instructions?
Yes. Every ISO toolkit includes a comprehensive set of user guides to help you implement effectively - even without prior consulting experience.
✅ Guides typically include:
Toolkit overview: Use cases, target users, structure, ISO scope, and application areas
How to use each document type: Purpose, real-life use cases, document control notes, and rollout steps
Customization guidance: How to adapt the templates for your business size, industry, certification scope, and internal policies
Workflow diagrams (if applicable): Showing logical connections among processes, templates, forms, and records
Recommended implementation sequence: Step-by-step instructions for deploying by clause, process, department, or implementation phase
🎯 Our goal is not just to provide professional ISO templates, but to ensure you know how to apply them effectively - with or without external consultants.
11. Are templates within one ISO toolkit duplicated across other toolkits?
No, contents are not simply duplicated. Each toolkit on ISO-Toolkits.org is purpose-built for a specific ISO standard, management system, industry need, or implementation objective, ensuring that documents remain relevant and practical.
✅ How we ensure content uniqueness:
Each toolkit is centered around a specific standard or management system theme, such as ISO 9001, ISO 14001, ISO 45001, ISO 27001, ISO 22000, ISO 13485, ISO 22301, or integrated management systems.
Every template is written with specific clauses, risks, controls, operational processes, and audit requirements of that standard in mind.
The structure, fields, records, metrics, and responsibilities in each file are tailored to distinct implementation and certification needs.
Example:
A "Risk and Opportunity Register" in an
ISO 9001 Quality Management Toolkit is different from an "Environmental Aspects and Impacts Register" in an
ISO 14001 Toolkit or a "Hazard Identification and Risk Assessment" document in an
ISO 45001 Toolkit.
🎯 This standard-based structure enables users to combine multiple ISO toolkits without unnecessary content redundancy, creating a more comprehensive and integrated enterprise management system.
12. Can I purchase only specific parts or individual sections of an ISO toolkit?
By default, our ISO toolkits are offered as complete, full-featured packages to ensure:
Logical consistency and completeness across the full ISO implementation process
A holistic view of the management system, from planning and operation to audit, review, and improvement
Avoiding gaps or missing critical templates by purchasing only partial content
🔄 However, in special cases, we can support:
Providing individual modules or sections (e.g., only internal audit documents, risk management templates, corrective action forms, management review materials, or training slides)
Helping you select a tailored bundle based on your certification stage, industry, audit findings, or short-term implementation needs
📩 Please contact our support team via email or the contact form for a custom quote or to request a sample preview before making a decision.
13. What payment methods are accepted?
We support multiple secure and globally accepted payment methods to accommodate customers worldwide.
✅ Accepted payment options include:
Credit/Debit Cards:
Visa, Mastercard, American Express, JCB, and other major cardsPayPal:
A secure and fast method for both individuals and businessesStripe:
Built-in checkout on our website, allowing direct card payments securelyBank Transfer (upon request):
For custom or bulk orders, we can provide manual bank transfer instructions as needed
🎯 Once payment is completed, you will automatically receive a confirmation email and secure download link within 15-60 minutes. If support is required, our team is available to assist promptly.
14. How will I receive the ISO toolkit after payment?
As soon as your payment is successfully completed, the system will automatically redirect you to a secure download page where you can immediately download the full ISO toolkit package.
✅ No need to wait for an email - the
download page appears
instantly after checkout.
✅ All files will be provided in a
single ZIP archive or through a
secure cloud-hosted link.
📌
Important Note:
Please ensure that your browser does
not block redirects after payment.
If for any reason you are not
redirected to the download page,
kindly contact us at
supports@iso-toolkits.org
- we will provide an alternative
download link promptly.
🎯 This instant delivery method ensures you receive the ISO toolkit quickly and securely, without delay.
15. Can I request an invoice or official billing document?
Yes. We can issue official invoices (electronic tax invoices) upon request for companies, organizations, or individuals who need to declare business expenses.
✅ How to request an invoice:
After completing payment, send an email to:
📩 supports@iso-toolkits.orgInclude the following details:
Company/organization name
Tax identification number (if applicable)
Billing address
Email to receive the invoice
Special notes (if any)
Order reference number or payment confirmation
Processing time:
Invoices are issued via email within 2-3 business days after we receive complete information.
📌 Invoices are issued by the official legal entity representing ISO-Toolkits.org and comply with tax and financial regulations applicable to international businesses.
16. Can I get support if I have trouble using the ISO templates?
Yes. We are committed to supporting our customers before, during, and after ISO toolkit implementation.
✅ Types of support available:
Email support:
Send questions to 📩 supports@iso-toolkits.org - we will reply within 24 business hours to help with usage, customization, or deployment.Implementation guidance:
We provide process flowcharts, usage notes, clause-based guidance, and recommendations to help users understand how to apply each group of templates.Customization advisory:
If your organization has unique industry, regulatory, operational, or structural needs, our team of experts can suggest how to tailor templates accordingly.Related materials recommendation:
If the current ISO toolkit doesn't fully meet your requirements, we can recommend complementary documents from our other toolkits.
🎯 Our mission goes beyond delivering templates - we aim to help you apply them effectively, achieve audit readiness, and drive continual improvement.
17. Who can I contact for advanced or specialized ISO support?
If you need expert-level support for using, customizing, or implementing our ISO toolkits in your organization, our team of consultants is ready to assist.
✅ Official support channels:
📩 Technical and content support:
supports@iso-toolkits.org
(Template usage, customization questions, ISO implementation guidance)
🛠 Specialized support may include:
Advisory on deploying ISO toolkit components based on your implementation roadmap, certification timeline, or audit findings
Template customization for specific industries such as banking, manufacturing, healthcare, education, logistics, construction, IT services, public sector, food, or medical devices
Full-scale implementation support for ISO certification, integrated management systems, internal audit programs, corrective action management, or continual improvement initiatives
🎯 We're not just a content provider - we are a trusted partner in helping you deliver successful ISO implementation and lasting management system value.
18. What if a file doesn't work or I have trouble opening it?
All files are thoroughly tested before release to ensure compatibility with major office software. However, in rare cases, users may encounter issues. Here's how to resolve them:
✅ Common issues and solutions:
File won't open or shows a format error:
→ Use Microsoft Office 2016+ or Google Workspace
→ Ensure the file was properly extracted if downloaded as a.zipExcel files show macro or content warnings:
→ Click "Enable Editing" and "Enable Content" to activate featuresMissing files or extraction issues:
→ Check your internet connection and re-download the file
→ Contact us if the issue persists - we'll provide a new download link
📩 If you face any technical error,
please email
supports@iso-toolkits.org
with a brief description and a
screenshot (if available).
👉 We are committed to resolving all
technical issues within
24 business hours.
Customer Reviews - ISO 17025 Toolkit
Verified customer feedback and implementation experiences for the ISO 17025 Testing and Calibration Laboratories Full Implementation Toolkit.
Related ISO Toolkits for Consultants provide ready-to-use templates, checklists, registers, reports, and implementation documents that help consultants deliver ISO projects faster, more consistently, and with greater confidence.
These toolkits support key consulting activities such as gap assessment, documentation development, control implementation, audit readiness, and continual improvement across a wide range of ISO standards.

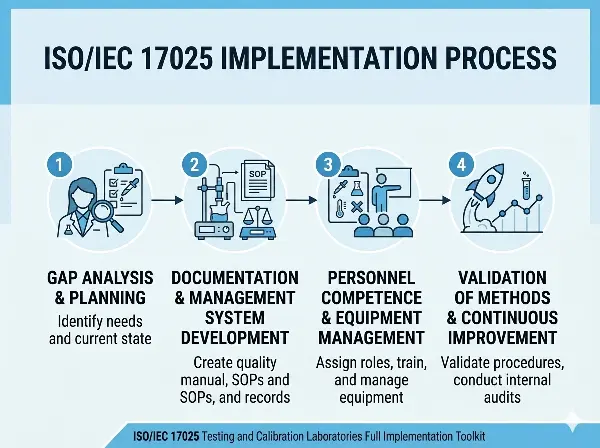
 Implementing requirements aligned with ISO/IEC 17025 can be complex and time-consuming, particularly for testing and calibration laboratories seeking to strengthen technical competence, ensure reliable results, and maintain consistent documentation across laboratory operations.
Implementing requirements aligned with ISO/IEC 17025 can be complex and time-consuming, particularly for testing and calibration laboratories seeking to strengthen technical competence, ensure reliable results, and maintain consistent documentation across laboratory operations.








The ISO Toolkit has helped us structure our implementation work clearly. It gave our team practical templates, organized procedures, and a reliable starting point for building our management system documentation.
After using the ISO Toolkit, our ISO preparation became much more organized. The documents are professional, easy to adapt, and helpful for aligning internal teams around clear compliance requirements.
Our consultants and internal managers found the toolkit very practical. It saved time, improved documentation consistency, and gave us a better framework for ISO implementation across departments.
The toolkit provides a strong foundation for ISO best practices. It helped us organize policies, procedures, records, and improvement actions in a way that is simple to maintain.
The ISO Toolkit brought structure to our compliance documentation and reduced the workload for our implementation team. It allowed us to focus more on improving processes instead of starting documents from scratch.
The ISO Toolkit is practical, well arranged, and easy to customize. It helped replace scattered files with a more complete document set for managing our ISO implementation activities.
The toolkit is very straightforward to use. It gave our team a clear implementation path, helped define responsibilities, and made ISO documentation easier for non-specialists to understand.
The ISO Toolkit gave us a better understanding of management system requirements and provided a user-friendly way to improve processes, controls, and internal documentation.
The toolkit helped me organize our ISO training, document review, and implementation planning. It made the entire preparation process more focused and easier to communicate with the team.
Excellent ISO Toolkit. It is highly useful for managers, consultants, and implementation teams who need practical documents to support ISO certification readiness.
A very useful toolkit and one of the most practical document sets I have used. It provides clear templates that can be adapted quickly for different ISO implementation needs.
These ISO Toolkits increased my confidence in managing implementation work. They helped us prepare documentation, assign responsibilities, and move toward a more mature management system.